Free ACT Science Practice Tests
ACT Science Practice Test 2
This is the second of our six free ACT Science practice tests. It’s designed to take your ACT Science prep a step further by helping you practice analyzing data, comparing experiments, and drawing logical conclusions. Review each question carefully, study the explanations, and keep building the skills that lead to a higher ACT Science score.
Passage II
When a force is applied to a gas confined in a container, the gas is compressed into a smaller volume. The pressure, P, exerted on the gas is equal to the applied force, F, divided by the area, A, of the surface on which the force acts, as shown in the equation:
P = FA
For a fixed amount of gas at constant temperature, the product of pressure and volume remains constant. This relationship can be expressed as:
P₁V₁ = P₂V₂
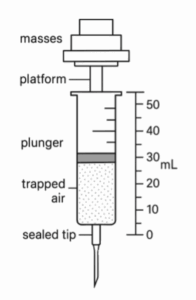
To investigate how externally applied forces influence the volume of trapped air, researchers performed two experiments that added mass to a gas syringe and measured the volume. The setup included a sealed syringe mounted vertically, a platform attached to the plunger for holding masses, and a scale on the syringe barrel to measure volume (see Figure 1).
Figure 1
Experiment 1
A syringe with a plunger of cross-sectional area 2.0 cm² was sealed at the tip. The plunger was positioned so that the trapped air had an initial volume of 50.0 mL at atmospheric pressure (101 kPa). Masses were added to the platform in increments, and the new volume was recorded after each addition. Table 1 shows the results.
Experiment 2
The procedure from Experiment 1 was repeated using a different syringe with a plunger of cross-sectional area 4.0 cm². The same masses were added, and the volume was recorded. Table 2 shows the results.